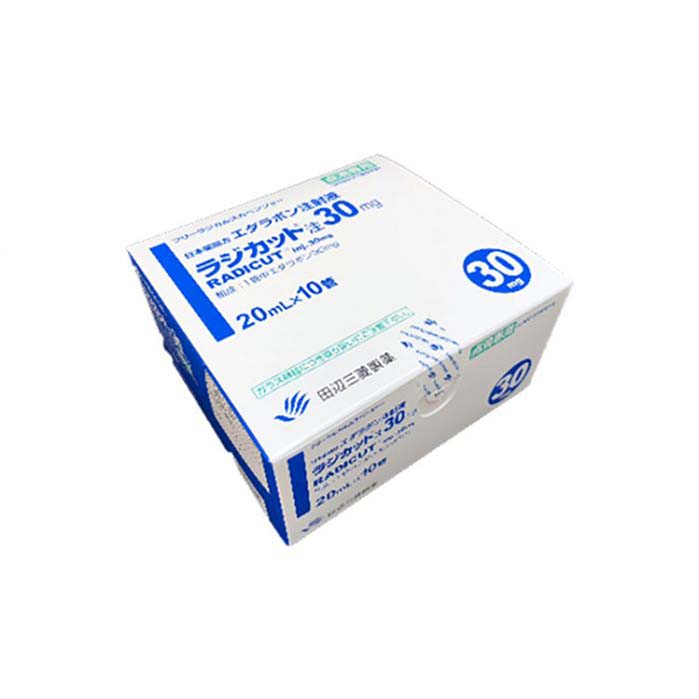
Buy Radicut (edaravone) Online For Sale
$882.00
Disease Indications: Amyotrophic Lateral Sclerosis (ALS), Cerebral Infarction
Manufacturer: Mitsubishi Tanabe Pharma Co., Ltd.
Usage: Intravenous
Medicine approved by:
Food and Drug Administration (FDA)
Health Canada
Pharmaceuticals and Medical Devices Agency (PMDA)
Radicut (edaravone) is prescribed for patients with amyotrophic lateral sclerosis (ALS) and acute ischemic stroke. It shares the active ingredient edaravone with Radicava but is marketed under a different brand name. At everyone.org, we provide Radicut (edaravone) to patients in need.
What is Radicut (edaravone) for?
Radicut (edaravone) is prescribed for the treatment of amyotrophic lateral sclerosis (ALS), also known as Lou Gehrig’s disease.
How does Radicut (edaravone) work?
To protect neurons from oxidative stress that may cause cellular damage, Radicut (edaravone) scavenges free radicals. This mechanism is expected to be effective in patients with acute cerebral infarction as well as those with ALS. One of the primary free radicals, hydroxyl radical (-OH), plays a significant role in the development of cerebral vascular disorder resulting from ischaemia. During ischaemia or reperfusion, the production of free radicals increases, which can lead to cell membrane injury and, ultimately, to cerebral dysfunction. While the etiology and disease progression of ALS are unknown, a possible involvement of oxidative stress caused by free radicals is suggested. Although the mechanism of how edaravone exerts its therapeutic effect in patients with ALS is not fully understood, it is expected to suppress disease progression by inhibiting oxidative damage to nerve cells.
How is Radicut (edaravone) taken?
Edaravone, marketed under the brand name Radicava or Radicut, is administered through daily intravenous infusion in a clinic or hospital setting. The treatment period consists of two cycles, each lasting for 14 days with a 14-day drug-free interval. During each cycle, patients will receive edaravone once a day for a total of 10 to 60 minutes. The dosage is determined based on a patient’s body weight with a maximum dose of 60 mg per infusion.
Are there any known side effects of Radicut (edaravone)?
Yes, there are known side effects of edaravone (Radicava/Radicut). Some of the most common side effects include bruising, gait disturbance, headache, muscle weakness, rash, back pain, and swelling. Other less common but serious side effects may include an allergic reaction, reduced kidney function, and shortness of breath. Additionally, edaravone may also increase the risk of developing infections. Patients are advised to consult with their healthcare provider and report any side effects experienced during treatment.
Clinical trials about Radicut (edaravone)
In order to obtain approval for the treatment of ALS, a study (MCI186-19) comparing the efficacy of edaravone with a placebo was conducted on 137 ALS patients. Of the 137 patients, 69 received edaravone while the remaining 68 received a placebo. The six-cycle study relied on the ALS Functional Rating Scale-Revised (ALSFRS-R) to compare the effects of the two treatments based on changes in the scores before and after treatment. The ALSFRS-R scale is a 12-item questionnaire which assesses fine motor, gross motor, bulbar, and respiratory function in patients with ALS during activities such as speech, salivation, swallowing, handwriting, and mobility, among others. Each item is scored from 0 to 4, with higher scores indicating better functional ability. The patients included in the study had ALSFRS-R scores of at least 2 on all 12 items, normal respiratory function, and a disease duration of no more than 2 years. The majority of patients (90%) in both groups received riluzole during the course of the study.
The results of the study showed that the decline in ALSFRS-R scores from baseline after 24 weeks was significantly lower in the edaravone-treated patients (Mean ± Standard Error (95 % CI) = – 5.01 ± 0.64) compared to the placebo-treated group (Mean ± Standard Error (95 % CI) = – 7.50 ± 0.66) with a difference of 2.49 (95 % CI=0.99, 3.98) on the ALSFRS-R scale; this represents a 33% difference between the groups. However, as with any medication, edaravone’s effect varies from patient to patient.
In December 2014, the European Commission granted orphan designation to edaravone for the treatment of ALS based on its expected mechanism of action. It is believed that motor neuron damage in ALS is caused by toxic molecules containing oxygen, and in some patients, a defect in the gene responsible for producing the enzyme called superoxide dismutase (SOD) leads to inflammation and nerve cell death. Edaravone is expected to act as an antioxidant and prevent nerve cell damage caused by oxygen-containing molecules, in addition to blocking the clumping of SOD in nerves, thereby reducing inflammation.
| Package | 10 bags of 30 mg/100 mL, 10 vials of 30 mg/20 mL |
|---|
Be the first to review “Buy Radicut (edaravone) Online For Sale” Cancel reply
Related products
Neurology
Neurology
Neurology
Neurology








Reviews
There are no reviews yet.