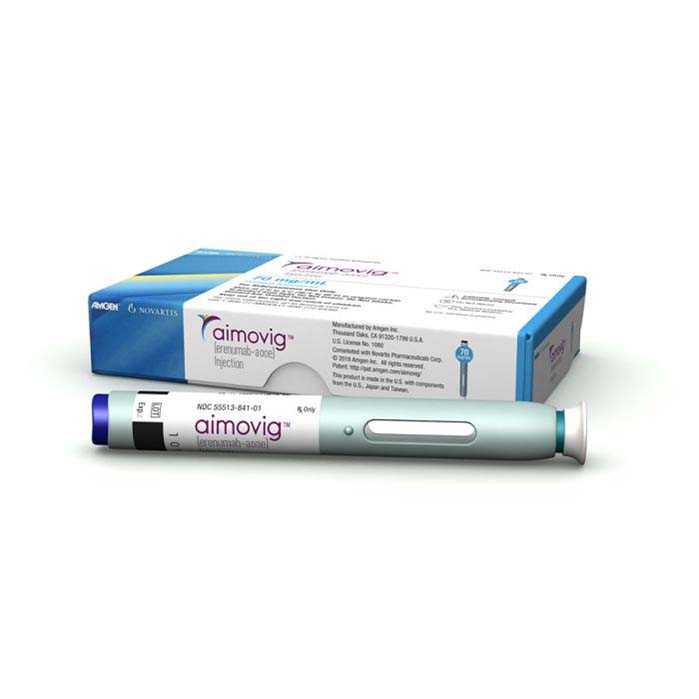
Buy Aimovig (erenumab) Online For Sale
From $411.00
DISEASE INDICATIONS: Migraine
MANUFACTURER: Amgen Inc. / Novartis Pharmaceuticals Corporation
USAGE: Subcutaneous
MEDICINE APPROVED BY:
European Medical Agency (EMA)
Food and Drug Administration (FDA)
Health Canada
Pharmaceuticals and Medical Devices Agency (PMDA)
Therapeutic Goods Administration (TGA)
Swissmedic (CH)
Medsafe (NZ)
Aimovig (erenumab) is a medication approved by the Food and Drug Administration (FDA) for the prevention of episodic or chronic migraines.
What is the purpose of Aimovig (erenumab)?
Aimovig (erenumab) is a prescription medication used for preventing migraines in adults. It belongs to a class of medications called calcitonin gene-related peptide (CGRP) inhibitors that work by blocking the activity of CGRP, a molecule that is involved in causing migraines. Aimovig is given as a subcutaneous injection (under the skin) once a month. It was approved by the US Food and Drug Administration (FDA) in 2018 for the prevention of migraine headaches in adults.
How does Aimovig (erenumab) work?
Aimovig (erenumab) is a monoclonal antibody medication that works by blocking the activity of a molecule called Calcitonin Gene Related Peptide (CGRP). CGRP is a small protein that is involved in the transmission of pain signals in the brain. Studies have shown that people with migraines have elevated levels of CGRP during an attack.
Aimovig works by blocking the receptors in the brain that CGRP uses to transmit pain signals, thereby reducing the levels of CGRP. This reduces the frequency and severity of migraines by preventing CGRP from triggering the inflammation that causes the migraine. By reducing the activity of CGRP, Aimovig helps to prevent migraines in people who suffer from them frequently.
How is Aimovig (erenumab) taken?
Aimovig (erenumab) is given as a subcutaneous injection which means it’s injected under the skin once a month. The recommended dose is 70 mg or 140 mg once a month, depending on the severity of your migraines and your response to the medication.
The injection is given using a prefilled autoinjector that you can use at home or with a prefilled syringe that you may need assistance with from a healthcare professional.
It’s important to follow the dosing instructions carefully and not to increase or decrease the dosage or frequency of the injections without consulting with a doctor. If you miss a dose, you should take it as soon as you remember, and then schedule your next injection at the regular time.
Are there any documented adverse effects associated with Aimovig (erenumab)?
Like all medications, Aimovig (erenumab) can cause unwanted side effects. Here are some of the most common side effects of Aimovig:
Common side effects:
– Injection site reactions, such as redness, swelling, pain, or itching at the injection site
– Constipation
– Muscle pain
– Joint pain
– Fatigue
– Flu-like symptoms
– Nausea
Less common but serious side effects may include:
– Allergic reactions such as rash, hives, itching, difficulty breathing or swallowing, or swelling of the face, lips, tongue, or throat
– Severe constipation or gastrointestinal obstruction.
– Liver damage
– High blood pressure
It’s important to tell your doctor if you experience any side effects while taking Aimovig. If you have a history of allergic reactions, liver problems, or problems with high blood pressure, you should inform your doctor before taking Aimovig.
Clinical trials
Several clinical trials have been conducted on Aimovig (erenumab) to assess its efficacy and safety for the prevention of episodic or chronic migraines. Here are some of the key clinical trials:
1. Study 1: A randomized, multi-center, 6-month, placebo-controlled, double-blind study conducted on 955 patients with a history of episodic migraine. The study concluded that Aimovig was effective in reducing the number of migraine days per month and was well-tolerated.
2. Study 2: A randomized, multi-center, 3-month, placebo-controlled, double-blind study conducted on 577 patients with a history of episodic migraine. The study showed that Aimovig was effective in reducing the number of migraine days per month and safe to use.
3. Study 3: A randomized, multi-center, 3-month, placebo-controlled, double-blind study conducted on 667 patients with a history of chronic migraine. The study revealed that Aimovig was effective in reducing the number of migraine days per month and was well-tolerated.
4. LIBERTY: A randomized, double-blind, placebo-controlled study conducted on 246 patients who have failed two to four preventive medication treatments for chronic migraines. The study found that Aimovig was effective in reducing the number of monthly migraine days and was generally well-tolerated.
5. STRIVE: A randomized, double-blind, placebo-controlled study conducted on 955 patients with a history of episodic migraines. The study revealed that Aimovig was effective in reducing the number of monthly migraine days, and was well-tolerated, with few adverse effects.
Overall, these clinical trials suggest that Aimovig is an effective and safe treatment for the prevention of migraine attacks in both patients with episodic and chronic migraines. However, consult with your healthcare provider to assess whether Aimovig is suitable for your specific condition.
| Package | 1 Autoinjector(s) of 70mg/ml, 3 Autoinjector(s) of 70mg/ml, 1 Autoinjector(s) of 140mg/ml, 3 Autoinjector(s) of 140mg/ml |
|---|
Be the first to review “Buy Aimovig (erenumab) Online For Sale” Cancel reply
Related products
Neurology
Neurology
Neurology
Neurology









Reviews
There are no reviews yet.