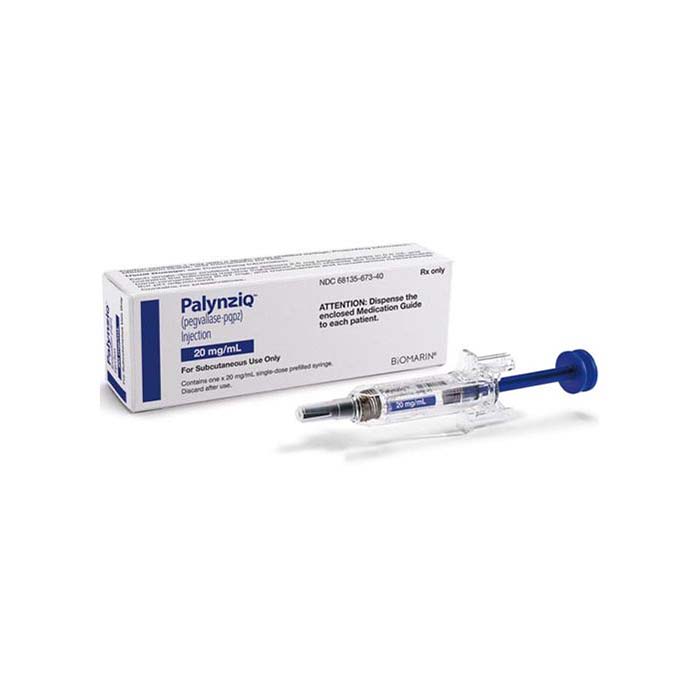
Buy Palynziq (pegvaliase-pqpz) Online For Sale
From $300.00
DISEASE INDICATIONS: phenylketonuria (PKU)
MANUFACTURER: BioMarin Pharmaceutical Inc.
USAGE: Subcutaneous
MEDICINE APPROVED BY:
European Medical Agency (EMA)
Food and Drug Administration (FDA)
Palynziq is an enzyme replacement therapy that has been developed for treating phenylketonuria (PKU), a genetic disorder that interferes with the body’s ability to break down phenylalanine. Palynziq introduces a modified version of the enzyme phenylalanine ammonia lyase (PAL) to break down phenylalanine and is administered as a subcutaneous injection on a daily basis.
Palynziq (pegvaliase-pqpz) is a breakthrough treatment for phenylketonuria (PKU), a genetic disorder that affects the body’s ability to break down an amino acid called phenylalanine (Phe). When left untreated, PKU can cause severe developmental delays, intellectual disabilities, seizures, and other serious medical problems. Palynziq was approved by the US Food and Drug Administration (FDA) in May 2018.
Palynziq is a form of enzyme replacement therapy that replaces a missing or deficient enzyme in the body. In people with PKU, the enzyme phenylalanine hydroxylase (PAH) is missing or not functioning properly. PAH is responsible for breaking down Phe into other amino acids that the body can use. Without PAH, Phe levels can build up in the bloodstream and cause damage to the brain and nervous system.
Palynziq works by introducing a modified version of the enzyme phenylalanine ammonia lyase (PAL) into the body. PAL is an enzyme that normally breaks down Phe in plants and bacteria, but it is not naturally produced by humans. The modified version of PAL in Palynziq has been engineered to closely resemble human enzymes, making it more effective and less likely to cause an immune response.
Palynziq is administered as a subcutaneous injection once a day. It comes in a prefilled syringe that patients can self-administer after receiving training from a healthcare provider. To reduce the risk of an immune response, Palynziq is dosed gradually and requires careful monitoring of Phe levels.
Clinical trials have shown that Palynziq is highly effective at reducing Phe levels in people with PKU. In a pivotal Phase 3 trial, patients who received Palynziq had significant reductions in Phe levels compared to those who received a placebo. Palynziq also improved the neurocognitive performance of patients, reduced the frequency of PKU-related symptoms, and increased dietary flexibility.
Palynziq is not without side effects. The most common side effects are injection site reactions, allergic reactions, and arthralgia (joint pain). Palynziq can also cause an increase in liver enzymes, which requires monitoring.
Palynziq has been described as a landmark drug for PKU because it represents the first significant advancement in PKU treatment in decades. Prior to Palynziq, the only treatment option for PKU was a strict low-Phe diet. While effective at reducing Phe levels, the diet is difficult to follow and can cause social and emotional isolation, among other challenges.
Palynziq offers a new, targeted treatment option that reduces Phe levels and improves the quality of life for people with PKU. It provides a sense of freedom and flexibility that many PKU patients have never experienced before.
In conclusion, Palynziq is an innovative and life-changing treatment for PKU. It represents a significant advance in the field of rare disease treatment and offers hope to the thousands of people with PKU who have struggled with the disease for years. Palynziq is a testament to the power of scientific research and the tireless efforts of researchers, pharmaceutical companies, and patient advocates who work together to improve human health and bring new treatments to those in need.
| Package | 1 single-dose prefilled syringe of 2.5 mg/0.5 mL, 1 single-dose prefilled syringe of 10 mg/0.5 mL, 1 single-dose prefilled syringe of 20 mg/1 mL, 10 single-dose prefilled syringes of 20 mg/1 mL |
|---|
Be the first to review “Buy Palynziq (pegvaliase-pqpz) Online For Sale” Cancel reply
Related products
Endocrinology
Endocrinology
Endocrinology
Endocrinology
Endocrinology
Endocrinology
Endocrinology
Endocrinology






Reviews
There are no reviews yet.